报告题目:Novel Synthetic Pathway for Multicyclic Polymers
报告人:Toshifumi Satoh教授(日本北海道大学)
报告时间:2019年8月28日, 9:30-10:30
报告地点:延长校区 平板显示中心二楼大会议室
报告摘要:Macromolecules possessing cyclic architectures have attracted much attention because of their interesting properties due to the absence of the polymer chain ends1. Therefore, multicyclic polymers, such as cage-shaped, 8-shaped and trefoil-shaped polymers, have been synthesized to investigate their unique properties. However, the synthesis of multicyclic polymers still remains challenging because of the complicated structures. Here, we propose a new synthetic strategy to multicyclic polymers, which involves the intramolecular consecutive cyclization of end-functionalized star polymer precursor with polymerizable groups. In this work, we attempted the synthesis of multicyclic poly(ε-caprolactone)s (cage- and multifoil-shaped PCLs) by the intramolecular consecutive ring-opening metathesis origomerization (ROMO) of star-shaped PCL with norbornenyl groups at the ω-chain ends (s-(PCLNB)x) and NB-(PCL-NB)y) (Scheme).
The star-shaped PCL with norbornenyl groups at ω-chain ends (s-(PCL-NB)3; Mn,NMR = 5,890 g mol–1, Mn,SEC = 9,440 g mol–1, Đ = 1.07) was synthesized by the ring-opening polymerization of ε-caprolactone using trimethylolpropane, followed by the condensation reaction with exo-5-norbornene-2-carboxylic acid (NB-COOH). The intramolecular consecutive ROMO of s-(PCL-NB)3 was carried out using the Grubbs catalyst 3rd generation (G3) in CH2Cl2 to lead cage-shaped PCL3. The NMR and MALDI-TOF MS measurement revealed that the fully proceeding of cyclization reaction and the formation of desired product, respectively. Furthermore, the SEC trace of the obtained product shifted to lower molecular weight region (Mn,SEC = 6,770 g mol–1, Đ = 1.09), indicating the formation of the cyclic product with smaller hydrodynamic volume as compared to s-(PCL-NB)3. Thus, these results confirmed that desired cage-shaped PCL3 was obtained by the intramolecular consecutive ROMO. In an analogous manner, varied arm-numbered cage-shaped PCLs and multifoil-shaped PCLs, were successfully synthesized using corresponding polymer precursors. Each multicyclic polymers could be prepared with controlled molecular weight.
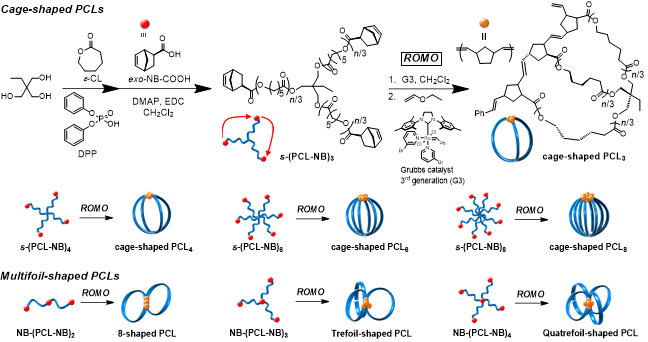
Scheme. Synthetic pathway for multicyclic PCLs
References
1. T. Yamamoto, Y. Tezuka, Soft Matter., 11, 7458-7468 (2015)